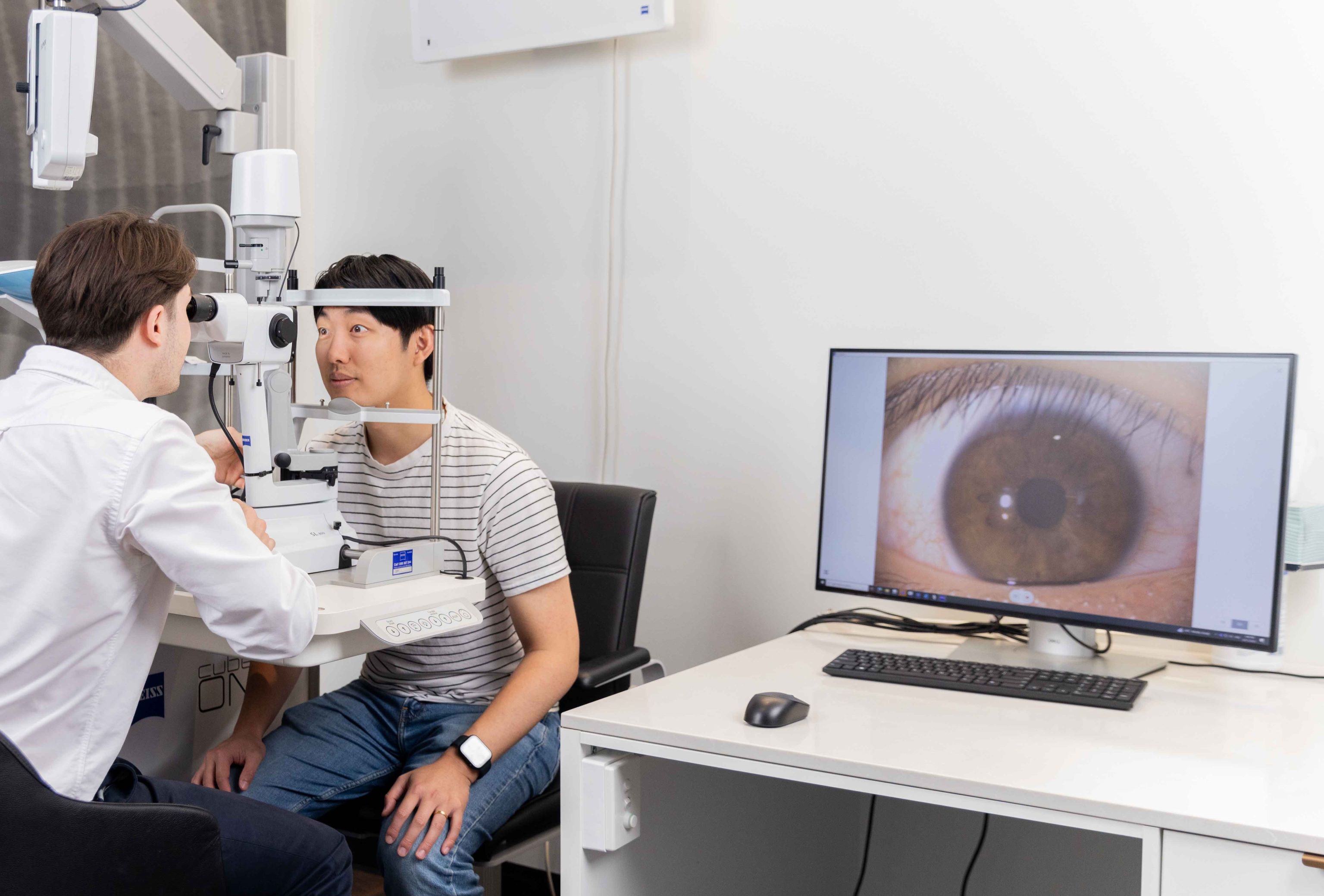
Fundus Autofluoresence
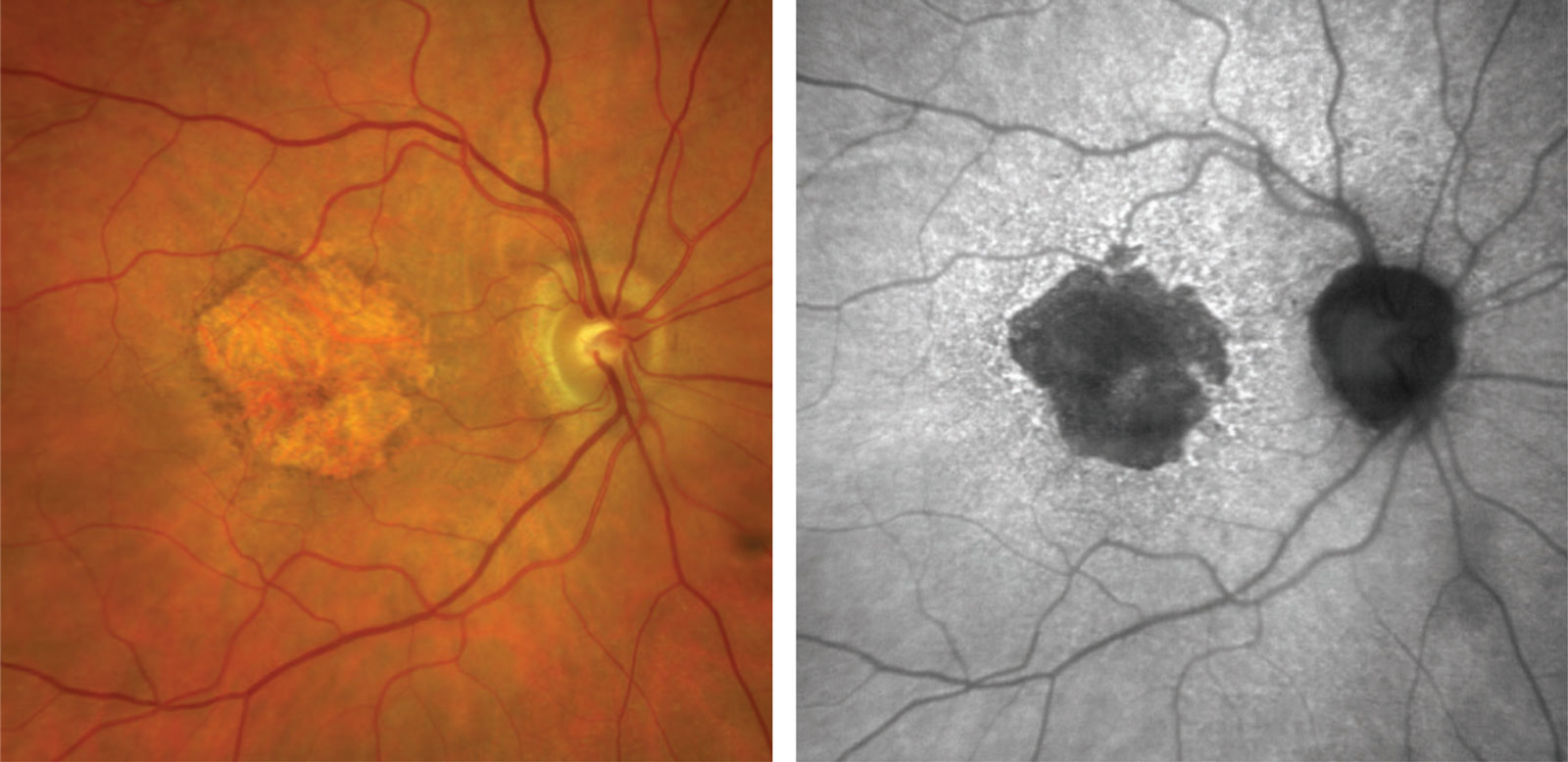
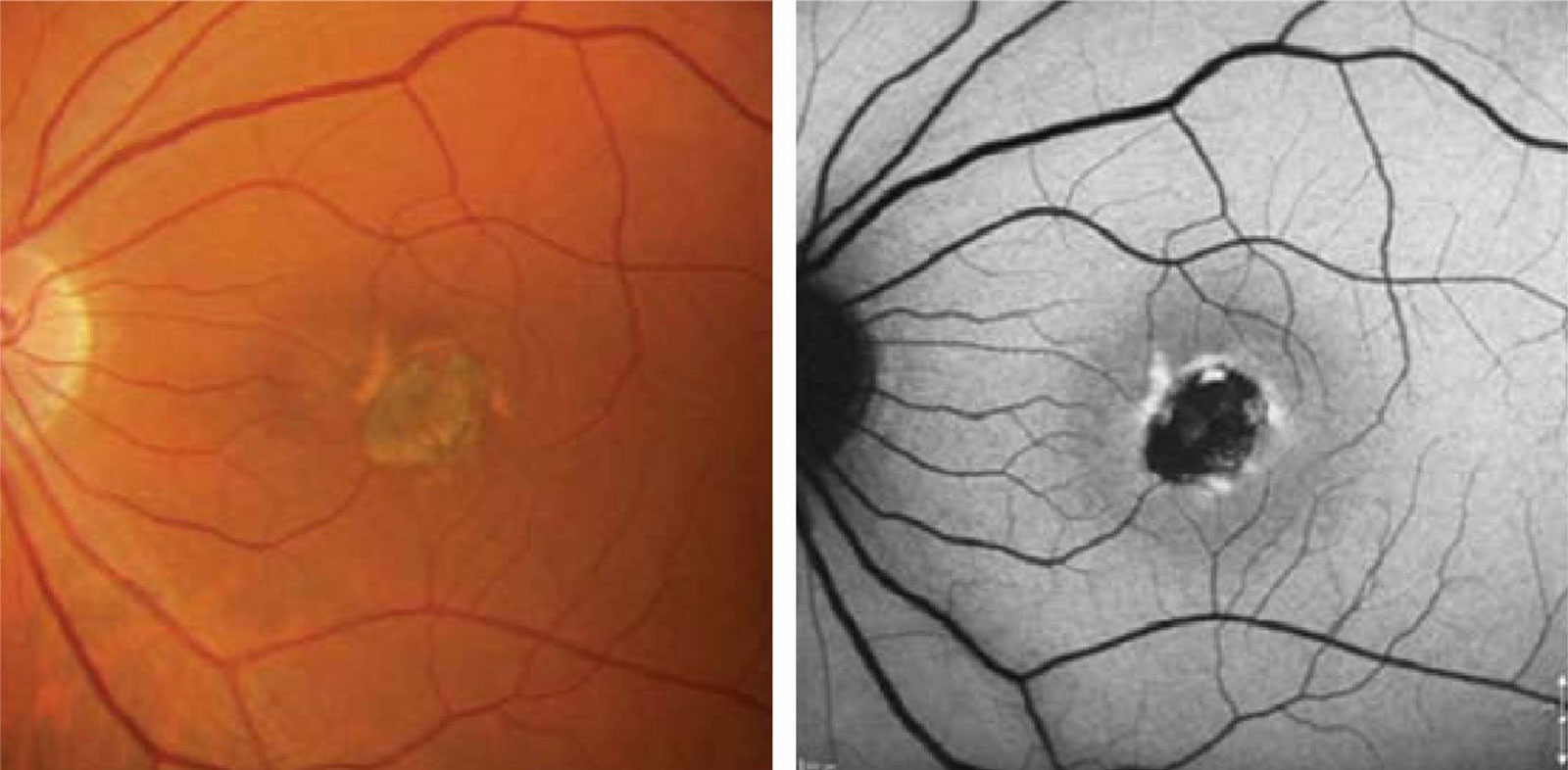
Fundus autofluorescence (FAF) is a non-invasive, high-definition scan of the deeper retinal layers at the back of the eye. FAF highlights areas of metabolic stress in detail that is above and beyond that of conventional retinal cameras.
Innovative Eye Care uses the top of the range Heidelberg Blue laser autofluorescence to take high-definition images of the retina. This offers diagnostic insights into a number of retinal conditions, including macular degeneration, Plaquenil toxicity, and hereditary diseases.
What is FAF?
The retina contains a metabolic indicator called lipofuscin, commonly referred to as the “wear and tear” pigment.1, 2 When light of 488nm is shone onto the retina, lipofuscin fluoresces and the amount of fluorescence corresponds with the amount of lipofuscin present.1 A number of hereditary and complex retinal diseases (including macular degeneration, medication toxicity and hereditary retinal disease) have characteristic increases or decreases in the amount of lipofuscin present in the retina.2, 3, 4 FAF provides detailed analysis of these conditions that allow the optometrists at Innovative Eye Care to accurately monitor your retinal health and potential prevent irreversible vision loss. This can be supplemented with other investigations including automated visual fields and OCT.5,6,7
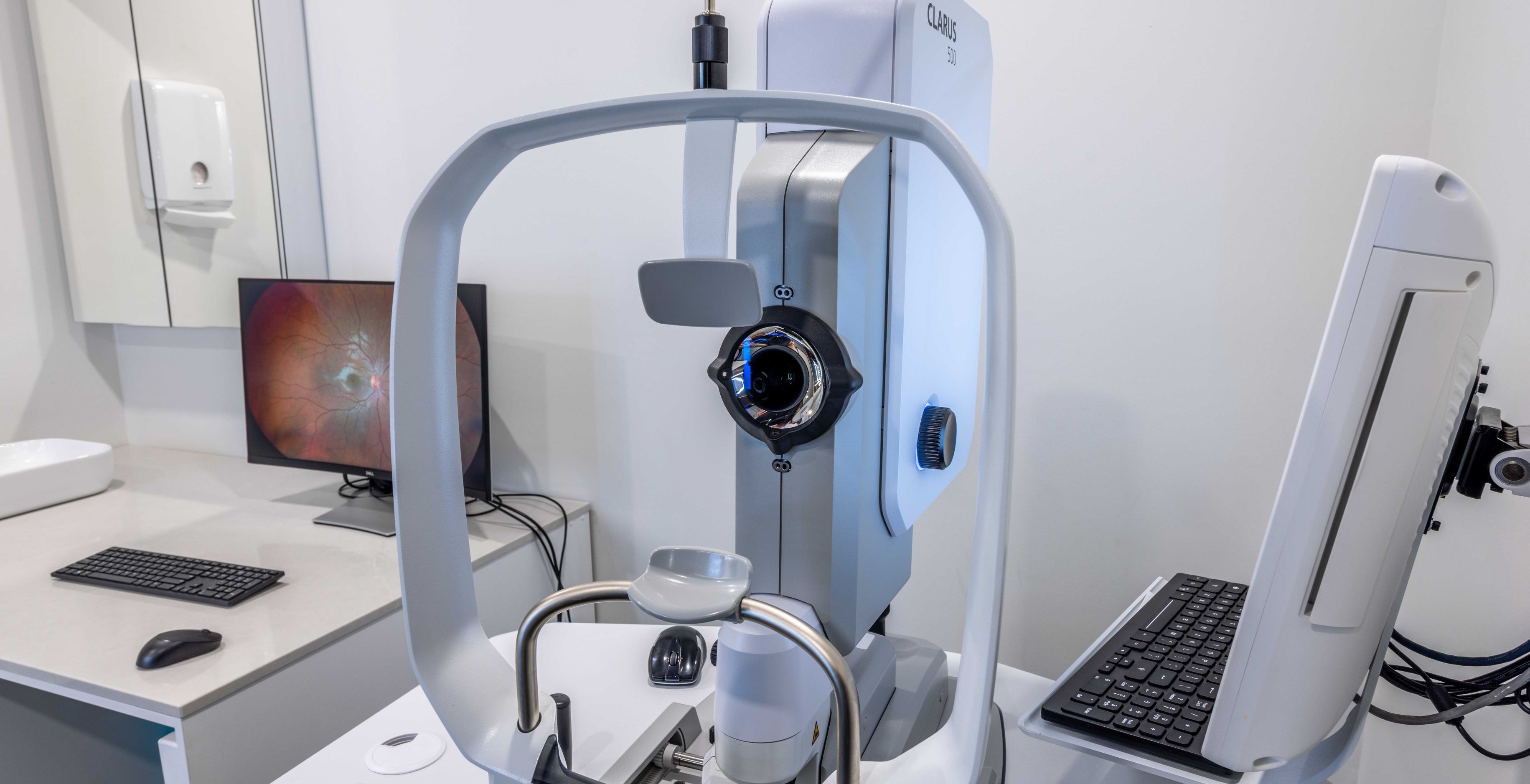
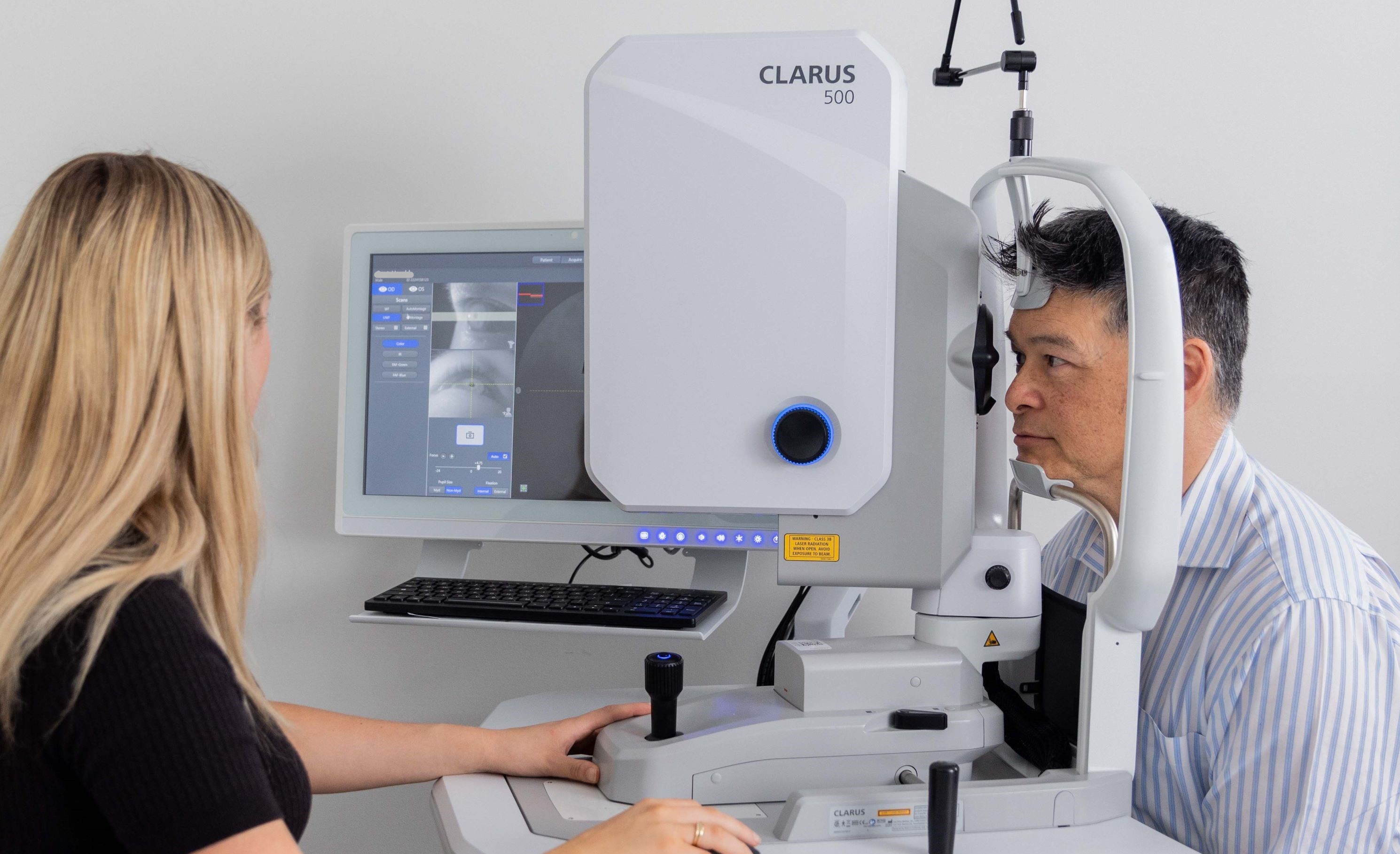
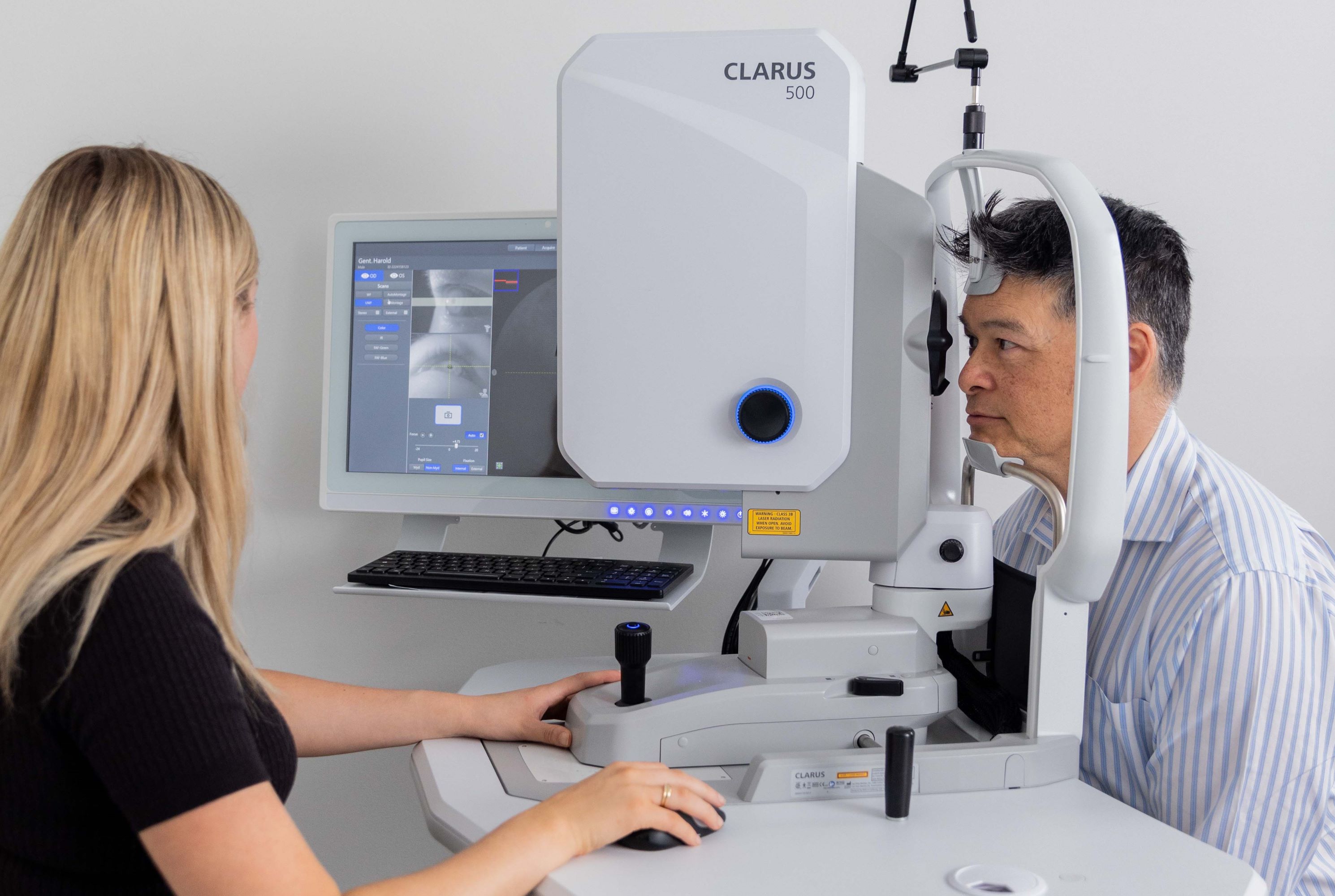
Zeiss Clarus
Heidelberg Spectralis
References
1) Lois N, Forrester JV. Fundus Autofluorescence. Philadelphia: Lippincott Williams & Wilkins; 2009.
2) Amin, XU. Changzheng, Chen. (2018).“Clinical application of ultra-wide field fundus auto fluorescence imaging". Chinese Journal of Ocular Fundus Diseases. 34(1): 89-92.
3) Frampton, GK. Kalita, N. Payne, L.Colquitt, J. Loveman, E. (2016). “Accuracy of fundus auto fluorescence imaging for the diagnosis and monitoring of retinal conditions: a systematic review. "Health Technol Assess. 20(31).
4) Theelen T, Berendschot TT, Boon CJ, HoyngCB, Klevering BJ. (2008). “Analysis of visual pigment by fundus autofluorescence.” Exp Eye Res.86:296-304.
5) Keel, S. et al. (2017). “Prevalence ofAge-Related Macular Degeneration in Australia”. Jama Ophthalmol.135(11):1242-1249.
6) Practice management in the era of anti-VEG therapy. Ophthalmology Time Europe; 2006(2)4:S1-8.
7) Marmor, M. Kellner, U. Lai, T. Melles, R.Mieler, W. (2016). “Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision)”. American Academy of Ophthalmology. 123(6): 1386-1394.
8) Lois N, Forrester JV. Fundus Autofluorescence. Philadelphia: Lippincott Williams & Wilkins; 2009.
9) Amin, XU. Changzheng, Chen. (2018).“Clinical application of ultra-wide field fundus auto fluorescence imaging". Chinese Journal of Ocular Fundus Diseases. 34(1): 89-92.
10) Frampton, GK. Kalita, N. Payne, L.Colquitt, J. Loveman, E. (2016). “Accuracy of fundus auto fluorescence imaging for the diagnosis and monitoring of retinal conditions: a systematic review.”Health Technol Assess. 20(31).
11) Theelen T, Berendschot TT, Boon CJ, HoyngCB, Klevering BJ. (2008). “Analysis of visual pigment by fundus autofluorescence.” Exp Eye Res.86:296-304.
FAQs
Please browse through some of our most frequently asked questions on this topic.
Speak to our friendly team today
Book your appointment now for personalised eye care tailored just for you.